This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.
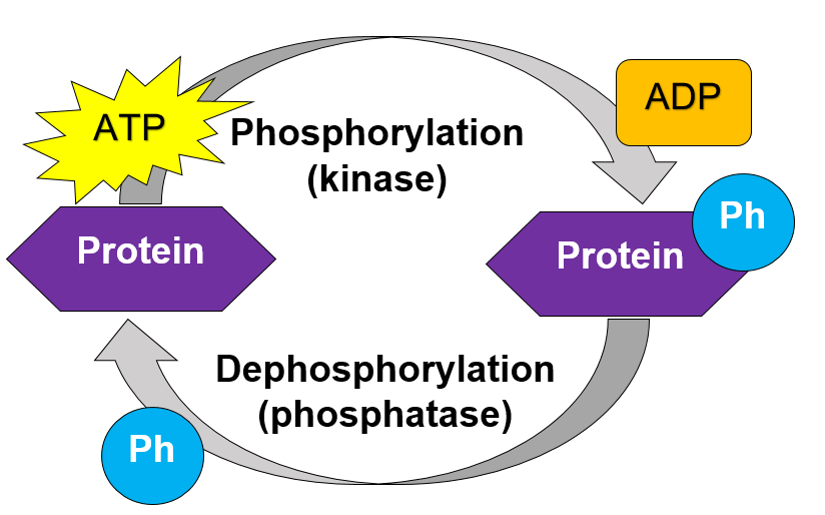
What is phosphorylation?
|
Protein phosphorylation is a mechanism used to essentially turn on and off protein interactions. It acts as a light switch for protein-protein interactions so that the cell can decide when to use the interactions and when not to. A kinase will add a phosphate group onto a protein at one of three specific amino acid sites, using ATP. The phosphoprotein will now be able to interact with other proteins in a way that it could not before. That is because the addition of the phosphate ion modifies the structure of a protein in such a way that it's properties are altered. In order to dephosphorylate, or turn off, a protein, a phosphatase removes the phosphate ion from the protein returning it to it's original form.
|
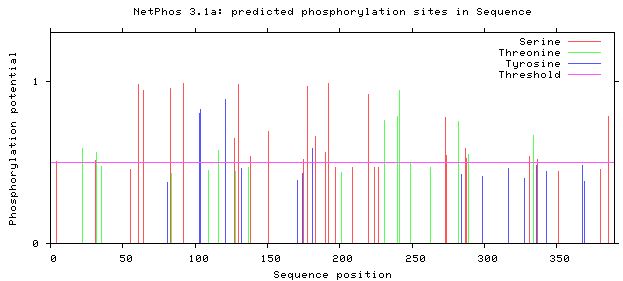
Where is TGFB1 predicted to be phosphorylation?
The three amino acids that are phosphorylated in human proteins are serine, threonine, and tyrosine. Each predicted phosphorylation site is one that has a line above the purple threshold line in the image below. The sites are only predicted because the data is derived from a protein in vitro, meaning in a dish. We can't be certain that these sites are also all phosphorylated in vivo, which means in a living organism.
We can see that most of the phosphorylation sites exist in the preprotein domain of TGFB1. This makes sense because, in addition to this domain being much larger than the TGFB domain, the preprotein domain helps the protein move around the cell and eventually become an excreted protein. It's to be assumed that many cascade pathways are involved in this delicate of a process, and thus, many more phosphorylation sites.
We can see that most of the phosphorylation sites exist in the preprotein domain of TGFB1. This makes sense because, in addition to this domain being much larger than the TGFB domain, the preprotein domain helps the protein move around the cell and eventually become an excreted protein. It's to be assumed that many cascade pathways are involved in this delicate of a process, and thus, many more phosphorylation sites.
Analysis
The preprotein domain of TGFB1 is more highly phosphorylated than the TGFB domain of the protein. This helps to support the ontology definition that the protein localizes in the extracellular space, and it also provides support for TGFB1 as a cytokine. Cytokines are excreted messengers and the role of the preprotein domain is necessary to the cytokine function of TGFB1.
References
[1] Sequence- and structure-based prediction of eukaryotic protein phosphorylation sites.
Blom, N., Gammeltoft, S., and Brunak, S.
Journal of Molecular Biology: 294(5): 1351-1362, 1999.
Images
[1] http://allswalls.com/apple-tree-branches.html
Blom, N., Gammeltoft, S., and Brunak, S.
Journal of Molecular Biology: 294(5): 1351-1362, 1999.
Images
[1] http://allswalls.com/apple-tree-branches.html