This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.
Introduction
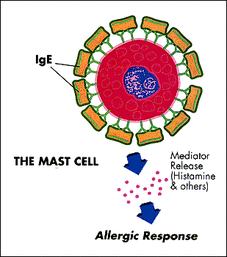
Allergic rhinitis, or hay fever, is a common disorder of the immune system. Symptoms of the disease include sinus swelling, pressure, and sneezing. At its best, it is a nuisance, but at its worst it can influence the quality of life of the patient. Allergic rhinitis is a hypersensitivity reaction. Antigens are proteins on the surface of a cell or molecule, and they interact with our immune cells. Antigens on an allergen, like pet dander, pollen or dust, interact with the immune system to cause a release of cytokines. Among these cytokines is TGFB1. In allergic rhinitis patients, TGFB1 is over expressed. [1] This inflammation ultimately leads to the inflammation that causes allergic rhinitis symptoms. [2]
Transforming growth factor beta isoform 1 (TGFB1) functions in several different systems throughout the body. We are focusing on its role as a cytokine in the immune system. A cytokine is a secreted molecule used for signaling. As such, TGFB1 stimulates a large number of immune cells to move around or take on a specific function. The over expression of TGFB1 in allergic rhinitis patients is problematic because the increase in the cytokine causes the immune system to over react to an otherwise harmless allergen. It would be beneficial to control the amount of TGFB1 expressed.
Transforming growth factor beta isoform 1 (TGFB1) functions in several different systems throughout the body. We are focusing on its role as a cytokine in the immune system. A cytokine is a secreted molecule used for signaling. As such, TGFB1 stimulates a large number of immune cells to move around or take on a specific function. The over expression of TGFB1 in allergic rhinitis patients is problematic because the increase in the cytokine causes the immune system to over react to an otherwise harmless allergen. It would be beneficial to control the amount of TGFB1 expressed.
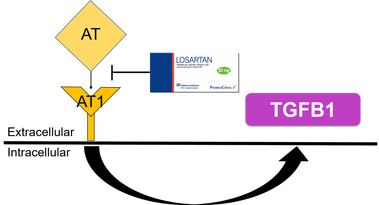
Losartan is an FDA approved medication used to treat high blood pressure. The drug acts on the angiotensin ii receptor pathway to decrease blood pressure, however it also decreases the amount of TGFB1 expressed. [3] It is unclear whether Losartan has the ability to treat the inflammation associated with allergic rhinitis by decreasing the levels of TGFB1. Because of TGFB1's inflammatory affects on the immune system, the over expression of the protein in allergic rhinitis patients, and Losartan's known ability to decrease levels of TGFB1, I hypothesize that Losartan will be able to effectively treat the inflammatory symptoms of allergic rhinitis by controlling expression of TGFB1.
The angiotensin ii receptor pathway is well conserved among species, as is the TGFB1 protein. Because of its numerous functions throughout the body, growth and development, this conservation is not surprising. It will be useful to compare the TGFB1 protein sequences among homologs with various types of immune systems, in order to try to narrow down which sequences are important for the immunologic function of the protein. I will use zebra fish (Danio rerio) to study the ability of Losartan to therapeutically decrease TGFB1 levels in the allergic response. Zebra fish have a well conserved copy of TGFB1 as well as the angiotensin ii receptor pathway. In addition, allergic inflammation can be induced by the addition of polysorbate 80 with hydrogen peroxide impurities to their tank water. [5] They may easily be treated with Losartan as well by the same method. A final advantage of using zebra fish to study Losartan's ability to treat allergic rhinitis is the ability to track neutrophils. There is a transgenic model of neutrophilic inflammation in zebra fish, which express GFP under a neutrophil specific marker. Because neutrophils are a key player in the early allergic response, it will be worthwhile and important for future studies to track their activities in this model of allergic rhinitis and Losartan treatment.
The angiotensin ii receptor pathway is well conserved among species, as is the TGFB1 protein. Because of its numerous functions throughout the body, growth and development, this conservation is not surprising. It will be useful to compare the TGFB1 protein sequences among homologs with various types of immune systems, in order to try to narrow down which sequences are important for the immunologic function of the protein. I will use zebra fish (Danio rerio) to study the ability of Losartan to therapeutically decrease TGFB1 levels in the allergic response. Zebra fish have a well conserved copy of TGFB1 as well as the angiotensin ii receptor pathway. In addition, allergic inflammation can be induced by the addition of polysorbate 80 with hydrogen peroxide impurities to their tank water. [5] They may easily be treated with Losartan as well by the same method. A final advantage of using zebra fish to study Losartan's ability to treat allergic rhinitis is the ability to track neutrophils. There is a transgenic model of neutrophilic inflammation in zebra fish, which express GFP under a neutrophil specific marker. Because neutrophils are a key player in the early allergic response, it will be worthwhile and important for future studies to track their activities in this model of allergic rhinitis and Losartan treatment.
Specific Aim 1
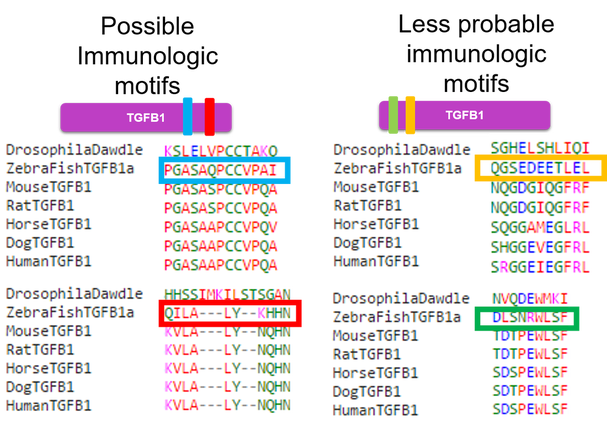
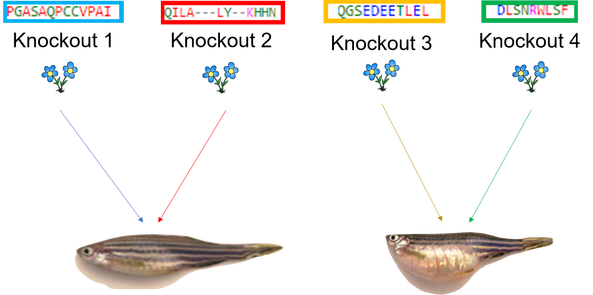
The main goal of aim 1 is to identify regions of the TGFB1 protein that are important for the immunologic function. First, I will use NCBI blast to identify homologs of the TGFB1 protein in several different species. I will make sure to include species that have both similar and dissimilar immune systems to our own. I will then use Clustal Omega to align the sequences. Based on the conserved regions between animals with similar immune systems and the negative support from the animals with dissimilar immune systems, I will be able to hypothesize about which regions may be important for immunologic function. I will confirm or refute these hypothesis and this method by using the CRISPR-Cas9 system to knock out the regions of the protein that I believe to be important or unimportant to the inflammatory response. I will then treat each knockout with an allergen. If I have knocked out a region of the protein that is essential to immunologic function, I expect to see introduction of an allergen without an inflammatory response. If I have knocked out a region of the protein that is not important to the immunologic function, I will expect to see inflammation upon introduction of the allergen.
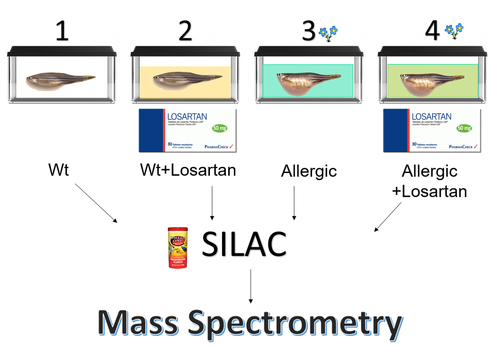
Specific Aim 2
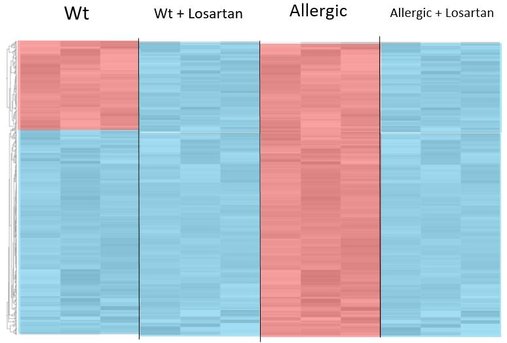
The main goal of aim 2 is to examine, through gene ontology, the differential gene expression patterns in allergic fish (treated with Losartan and untreated) compared to wild type fish (treated with Losartan and untreated). I will create four tanks for each scenario. Tank one will contain wild type fish, without the induced inflammatory response and without Losartan. Tank two will contain treated wild type fish, without the induced inflammatory response and with Losartan. Tank three will contain allergic fish, with the induced inflammatory response and without Losartan. Tank four will contain treated allergic fish, with both the induced inflammatory response and Losartan. I will then retrieve tissue samples from the same site on all of the fish, purify the mRNA, and sequence the transcriptomes of the four treatment groups. I will visualize the results using a heat map, and include genes with a 2 fold or more change in gene expression from the wild type without treatment. In the allergic untreated fish, I expect to see an increase in gene expression of ontology groups like inflammation or defense response. In the allergic treated fish, I expect to see more baseline (similar to wild type untreated) levels of gene expression in these categories because I am hypothesizing that Losartan will have the ability to treat inflammation. In both the treated wild type and treated allergic fish, I expect to see a decrease in expression of blood pressure maintenance genes. That is simply because Losartan is used to treat high blood pressure, so I expect a side effect of using it to treat allergies may be low blood pressure. Therefore the red top piece of the heat map in the wt represents blood pressure maintenance genes, and the bottom blue half represents immunological, inflammatory or defense response genes. Red is upregulation, blue is downregulation.
Specific Aim 3
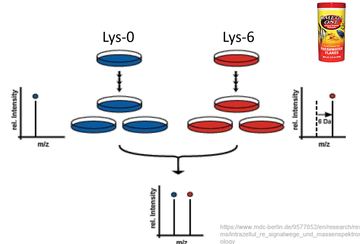
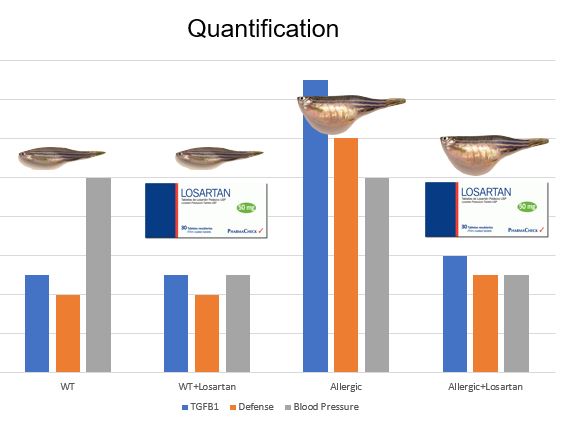
The main goal of aim 3 is to quantify the levels of proteins, like TGFB1, from the ontology categories that are upregulated or downregulated. I will establish four populations of the fish as described in aim 2. I will preform stable isotope labeling in vivo by feeding the fish with Lys-6 fish food. [6] I will preform reverse liquid chromatography and a mass spectrometry analysis. This will allow me to quantify proteins that are included in inflammatory, defense response, or blood pressure maintenance gene ontology categories. Using wild type as a baseline, I expect to see higher quantities of inflammatory proteins in untreated allergic fish. I expect to see baseline, or near baseline, quantities of inflammatory proteins in wt, wt + Losartan, and allergic + Losartan groups. In both groups treated with Losartan, I expect to see decreased quantities of blood pressure maintenance proteins.
Conclusions & Future Directions
Uncovering Losartan's ability to treat allergic rhinitis symptoms by controlling TGFB1 expression would be a huge step toward curing this extremely common affliction. Treatment of allergic rhinitis will improve the patients quality of life immensely, and the ability to treat with a drug already known to be safe for humans would speed the process. Following that logic, it doesn't seem like a huge leap to begin human drug trials with Losartan to treat allergic rhinitis. Precautions would have to be taken of course, as a decrease in blood pressure would be expected. A more rare side effect of Losartan is neutropenia, a severe decrease in or absence of neutrophils. This side effect would be an extremely interesting aspect of the drug in the future, because we know that there is a large presence of neutrophils during the early allergic rhinitis response. If Losartan can decrease that number, maybe it can treat allergic rhinitis in more ways than just by decreasing TGFB1. Because of the transgenic neutrophil marked line of zebra fish, this study could be of some use to future studies looking to investigate further into the ever changing world of allergic rhinitis treatment as we search for a cure.
Final Draft
| tgfb1presentationfinal_guerra_4.20.17.pdf | |
| File Size: | 1930 kb |
| File Type: | |
Draft 2
| tgfb1presentationdraft2_guerra_4.4.17.pptx | |
| File Size: | 2645 kb |
| File Type: | pptx |
Draft 1
| tgfb1presentationdraft1_guerra.pptx | |
| File Size: | 1453 kb |
| File Type: | pptx |
References
[1] Wan, Y. Y., & Flavell, R. A. 2007. “Yin-Yang” functions of TGF-β and Tregs in immune regulation. Immunological Reviews, 220, 199–213. http://doi.org/10.1111/j.1600-065X.2007.00565.x
[2] Debock I., Flamand V. 2014. Unbalanced Neonatal CD4+ T-cell Immunity. Frontiers in Immunology, 393. 1664-3224. https://doi.org/10.3389/fimmu.2014.00393
[4] Renshaw et al. (2006). A transgenic zebrafish model of neutrophilic inflammation. American Society of Hematology. 108:3976-3978. https://doi.org/10.1182/blood-2006-05-024075
[5] Yang et al. (2014). Tween-80 and impurity induce anaphylactoid reaction in zebra fish. Journal of applied Toxicology. 35,3:295-301. 10.1002/jat.3069
[6] Konzer et al. (2013). Stable Isotope Labeling in Zebrafish Allows in Vivo Monitoring of Cardiac Morphogenesis. Mol Cell Proteomics. 12(6): 1502-1512. 10.1074/mcp.M111.015594
Images
[1] http://www.cityallergy.com/13-the-process-of-an-allergic-reaction/
[2] Debock I., Flamand V. 2014. Unbalanced Neonatal CD4+ T-cell Immunity. Frontiers in Immunology, 393. 1664-3224. https://doi.org/10.3389/fimmu.2014.00393
[4] Renshaw et al. (2006). A transgenic zebrafish model of neutrophilic inflammation. American Society of Hematology. 108:3976-3978. https://doi.org/10.1182/blood-2006-05-024075
[5] Yang et al. (2014). Tween-80 and impurity induce anaphylactoid reaction in zebra fish. Journal of applied Toxicology. 35,3:295-301. 10.1002/jat.3069
[6] Konzer et al. (2013). Stable Isotope Labeling in Zebrafish Allows in Vivo Monitoring of Cardiac Morphogenesis. Mol Cell Proteomics. 12(6): 1502-1512. 10.1074/mcp.M111.015594
Images
[1] http://www.cityallergy.com/13-the-process-of-an-allergic-reaction/